Erroneous assumptions based on the 1958-1964 works of Bert Bolin are deep in the orthodoxy of human-CO2-caused global warming /climate change. Bert Rickard Johannes Bolin (1925 – 2007) was a Swedish meteorologist who served as the first chairman of the UN’s Intergovernmental Panel on Climate Change (IPCC), from 1988 to 1997. “Bolin is credited with bringing together a diverse range of views among the panel’s 3,500 scientists into something resembling a consensus.” (Sundt, Nick. 1995.) He was professor of meteorology at Stockholm University from 1961 until his retirement in 1990. Bolin’s papers were based on differential equations, calculus and assumed analogies in science. Bolin’s work omitted critical information and calculations.
Bolin’s math was elegant. And it is true, as he derived, that diffusion of CO2 gas and dissolved inorganic and organic carbon in the ocean water column is the rate limiting variable in vertical migration in the ocean water column.
The problems with Bolin are not in the calculations which he included, and which are referenced today in UN IPCC orthodoxy, as well as by many skeptics still today. The problems are the facts and math which Bolin omitted. Bolin derived and integrated the CO2 gas migration rate and dissolved carbonate reaction rates in the vertical water column in ocean. From there he derived the thickness of a thin layer of ocean surface that implies the observed slope of CO2 concentration (e.g., the Keeling curve) could not be CO2 emissions from the ocean. Bolin’s calculations showed the CO2 migration rate in ocean was too slow, and “chemical enhancement” (which were the reactions and rates of the series of carbonate reactions) was insufficient in his derived thin layer at the top of the vertical ocean water column.
And it is true that the CO2 gas migration is the rate limiting step in the vertical ocean water column. However, CO2 gas and dissolved carbon do not need to migrate horizontally or vertically in order to reverse react and produce CO2 gas emissions from ocean surface. The various carbonate ions and ionized CO2 gas are perpetually surrounded by water ions as well as calcium ions in ocean surface. Bolin considered in his calculations the thin layer as well as the well-mixed layer, the top 2 layers (~20 meters to ~100 meters) of the ocean water column. But, Bolin omitted ocean surface area. Using the chemical enhancement and migration rate in the vertical ocean water column in calm surface Bolin and Erik Eriksson (1959) concluded, “It is obvious that an addition of C02 to the atmosphere will only slightly change the C02 content of the sea but appreciably effect the C02 content of the atmosphere. It is possible to deduce a relation between the exchange coefficient for transfer from the atmosphere to the sea and the corresponding coefficient for the exchange between the deep sea and the mixed layer.” (Underlining and bolding by Bud)

The white chalk cliff of east Sussex, England
How then does ocean contain 40 to 50 times the amount of CO2 as air according to geology and ocean chemistry? What, for example, formed the famous white cliffs of Dover and Sussex, England and Normandy, France? That relation and those coefficients are expressed in Henry’s Law, Graham’s Law, and Fick’s Law. Those white cliffs were once CO2 in the air which was absorbed into the ocean and incorporated into sea life or precipitated as one of the forms of solid calcium carbonates such as limestone. Bolin was almost there. But, these laws apply at the gas-liquid exchange interface, that is sea surface and all water surface exposed to air, but not to deep sea and the well mixed middle layer, or even 10 centimeters below a calm surface, nor do they apply to the various atmospheric layers above the thin layer at the surface.
Bolin (1960) states, “The transfer of carbon dioxide through the atmosphere and the sea takes place by turbulent processes except possibly in the intermediate vicinity of the sea surface where molecular diffusion may play a role at least in the case of a smooth surface. The transfer across the sea surface is dependent on the number of molecules colliding with the interphase and being retained in the water when coming from the air and vice versa.”
Bolin omitted from his calculations (a) the phase-state equilibrium reaction that applies to non-ionized CO2 gas in ocean surface and (b) the several acid-base carbonate system equilibrium reactions that apply to ionized CO2 when both (a) and (b) are occurring in about 361 million square kilometers of ocean surface thin layer! Perhaps I am missing something in hidden derivation steps, but it appears Bolin only considered an ocean surface area of 2 X 10-5 cm2. I have not found any consideration of the horizontal surface area of the ocean in his papers, even though it is equally important to ocean thin layer thickness in calculating the diffusion rate of CO2 gas into or out of the ocean. This is a huge error of omission which UN IPCC orthodoxy uses to deny that the slope of CO2 concentration could be due to natural emissions from ocean surface.
Net diffusion flux of a gas through a surface is specified by Fick’s Law. The area of the gas exchange surface is equally important as the thickness of the exchange surface in Fick’s Law. Why did Bolin omit ocean surface area? Flux is the mass (or moles) of CO2 gas diffusing through a surface area in a unit of time, for example, gigatonnes of CO2 gas emitted per square kilometer per day.
On the other hand, Bolin’s calculations of turbulence in the vertical ocean water column leading to the rapid vertical migration rate of CO2 and dissolved carbonates, and eventual emission of CO2 gas into air are important parts of the explanation for the large effect of El Ninos on CO2 atmospheric emissions and rates of change of CO2 concentration. Bolin stated, “For a rough sea surface the transfer velocity [for the exchange of CO2, between the atmosphere and the sea] may be 10 to 100 times larger.” ..than a smooth surface. Turbulence increases both the temperature of the water and the surface area.
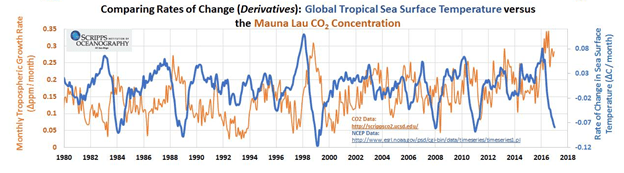
Changes in CO2 concentration rapidly follow changes in sea surface temperature as shown in the following graph.
References:
Bolin, Bert. 1960. On the Exchange of Carbon Dioxide between the Atmosphere and the Sea, Tellus, 12:3, 274-281, DOI: 10.3402/tellusa.v12i3.9402 To link to this article: https://doi.org/10.3402/tellusa.v12i3.9402
Bolin, Bert & Eriksson, Erik. 1959. Changes in the Carbon Dioxide Content of the Atmosphere and Sea due to Fossil Fuel Combustion. Pgs. 130-142. The Atmosphere and the Sea in Motion.
https://archive.org/details/atmosphereseainm0000boli
Sundt, Nick. 1995. IPCC Chairman Bert Bolin Builds Consensus. Center for Global Change. Archived from the original on 2002-11-16. Retrieved 2003-08-13. http://www.globalchange.org/profall/95jul16d.htm

Pingback: A conservative calculation of specific impulse for CO2 | budbromley
Pingback: A wake-up call for the „woke“ alarmed by the fake climate crisis - Climate- Science.press
Pingback: A wake up call for the “woke” alarmed by the fake climate crisis | budbromley
Bud,
Some more thoughts on carbonate oolites.
The study of ooilte formation using a hydrological test bed model would be a scientific endeavour of real interest and merit. The model would require a seawater tank with a calcium bicarbonate solution of known isotopic composition. Wave generating capabilities, an artificial beach seeded with calcite crystal grains, a controlled nitrogen atmosphere with air circulation to model the natural wind, a source of radiant heat (sunlight) and also appropriate thermometers, carbon dioxide gas detectors and isotopic composition determination capabilities.
I wonder if there would be anyone willing to undertake this?
LikeLike
Philip, after thinking on carbonate oolites for a bit, I am curious about the amount that might be present and exposed at the interface of air and water worldwide. If this is a significant amount, relative specifically to net Gt CO2 concentration in air, then this may be research that could find funding. If not, then are there any commercial uses for these carbonate oolites or specific types of carbonate oolites? (I assume that oolite mineralization occurs in various forms as do other carbonate precipitant versions, is that correct?)
LikeLike
Thanks Bud,
Oolites are an area of Carbonate Geology that gets left out of mainstream knowledge. It is commonly assumed that all carbonate rocks are organic in origin and this is false. There is the issue of the core seed crystal in the centre of the oolite being formed by bio-erosion, but the oolite formation process around a seed crystal core is similar to the formation of cloud droplets around a dust grain, and for similar precipitation/concentration reasons.
Oolites undergo the same erosion process as all other carbonate rock because they are a mono-mineralic substance (OK we have magnesium dolomites but they too can form as inorganic precipitates).
The key points for me are these:
1. The failure to observe and measure in the field the release of carbon dioxide gas into the atmosphere from coastal beach swash zones where oolites are actively forming. (This oolite sand grain generation is not a slow process).
2.The failure to measure the isotopic concentration of the carbon dioxide gas released into the atmosphere by the formation of swash zone oolites. This concentration ought from crystal fractionation first principles involve the preferential release of the light C12 carbon dioxide gas molecules directly into the air with the retention of the heavier C13 as carbonate in the lattice of the calcite crystals.
LikeLike
Thanks Bud for this well-written and easy to understand article.
Aloha, Larry
>
LikeLiked by 1 person
Bud,
One of the key features here from my perspective as a geoscientist is the role of carbonate oolite sand grains that are inorganically precipitated in the turbulent swash zone of beaches at the margins of warm tropical shallow seas that contain carbonate lagoons and ramps.
Oolite carbonate sand grains are onion ring structures (similar to hail stones in their mode of formation) and because they form from the inorganic precipitation of calcium bicarbonate solution release the produced carbon dioxide gas directly into the atmosphere without any intermediate biological intervention.
The quantity of oolitic carbonate sediments in the geologic record is not fully known, but a minimum estimate of 30% of all carbonate rocks being inorganically formed oolites is a reasonable supposition.
LikeLiked by 1 person
Aloha Philip. Thanks for this very interesting comment. Yet another source and sink of CO2 gas.
Can we assume weathering of carbonate oolites to cause release would require a specific energy activation profile and specific chemistry in the splashing waters at the beach? If you can put boundaries on those conditions, it might be possible to calculate when, where and how much carbonate oolites exist.
LikeLike
Pingback: The monumental CO2 error by IPCC’s first chairman - Climate- Science.press