CO2, water, and CH4 (methane) and some other minor gases are so-called “greenhouse gases” because they absorb infrared energy. However, be careful to avoid the common misunderstanding that the earth and its atmosphere behave like a garden greenhouse; that is not true. Greenhouses are closed systems except to incoming energy. Earth is an open system, open to receive energy, as well receive gases and other matter from space and also open to transmit energy, gases and matter back into space. A greenhouse is a poor analogy for earth’s climate.
Near earth’s surface, greenhouse gases re-emit almost instantly the energy they absorbed, emitting that energy at a slightly lower energy than which it was absorbed but emitting it into 3D space. Rarely is that emission mentioned in the news or by climate alarmists. Proponents of global warming want you to believe that energy is stored and builds up in the atmosphere in a sort of hothouse or pressure cooker, but that is not true.
In most locations, the concentration of water vapor (including clouds) in the atmosphere is far higher than CO2 and methane. That is, there are far more water vapor molecules in a given volume of air than there are CO2 or methane molecules. Near the equator and bodies of water, swamps and tundra, water vapor concentration in the air will be high. That is, the air is saturated, no more water vapor can be added to the air at that temperature and air pressure. Cold air holds very little water vapor. Cold air over cold water is also saturated, but air at the poles is usually very dry, water vapor concentration is very low. A volume of warm air holds much more water vapor than the same volume of cold air. Air in the tropics is usually humid.
The tropics and sub-tropics receive large amounts of solar radiation during the day, but none at night. During the day, the surface of ocean water is warmed by solar radiation and that warmth results in release of large amounts of CO2 and CH4 as well as water vapor into the atmosphere; the amount of these released gases dwarfs all emssions of these gases by humans. At night, the cooler ocean surface absorbs large amounts of CO2. CO2 is very soluble in cold water. A cold soda-pop keeps its bubbles.
The oceans and subsea floor hold far more methane (CH4) than all petroleum and fossil fuels ever discovered, and this methane is continuously degassed out of warm water and soil into the air. CH4 released into open air spontaneously converts (oxidizes) to CO2 and water at standard temperature and pressure. CH4 + 2 O2 -> CO2 + 2 H2O + a small amount of energy. Almost all CH4 originated as living cells. The cells died, entered the soil, rivers, lakes and ocean as runoff, metabolized and degraded by natural biological activity; this is the ultimate source of almost all fossil fuel on earth. That natural resource is continuously renewing and unlikely to be exhausted so long as there is life in the oceans.
Water vapor is the dominate greenhouse gas, absorbing by far both more direct solar energy and indirect solar energy re-emitted by the earth. More than 70% of the earth is covered with liquid water and about 40% of the earth and liquid water is receiving direct solar radiation every day. Additional liquid water is on land and in soil and plants. Solar radiation increases the molecular activity of liquid water molecules until they can no longer be held by the surface tension of the water. These active water molecules evaporate, i.e. release energetic water molecules, to become gaseous water vapor in the atmosphere. This release of energetic molecules cools the surface of the water, not warm the water, they just left. Your skin is cooled when your sweat evaporates.
Liquid water is far denser than air. Dense liquid water in the oceans is a huge reservoir of stored energy. The oceans contain greater than 1000 times more energy than the atmosphere; this means that earth’s atmosphere as a whole – regardless which gases make up the atmosphere – cannot warm the oceans in any significant amount. And this means the oceans are warming the atmosphere. The oceans are controlling the weather and climate on earth, not the atmosphere. The oceans are warmed by direct solar radiation as well as volcanic and tectonic forces resulting from gravity.

Figure 1
The specific gas molecules (nitrogen, oxygen, water vapor, as well as CO2, methane, and several other trace gases) that make up the atmosphere, each with their various concentrations, individually or in combination, are irrelevant to the long-term average temperature of the planet. In other words, an atmosphere of 100% CO2 could not significantly change the earth’s average temperature; instead, the ocean with its huge heat content would control the temperature of the 100% CO2 atmosphere. The atmosphere cannot warm significantly the earth over an extended time period, such as a year. But a warm ocean current, such as an El Nino, can change our climate and weather overnight.
However, water vapor and CO2 cause relatively minor temperature changes of the atmosphere at the surface (troposphere.) Water vapor and CO2 delay the release of energy from the earth into outer space, and this is most evident at night. Night time air temperatures at earth’s surface would be significantly cooler without water vapor and CO2. In a sense, greenhouse gases act as a nighttime insulator for the earth.
The effects of this are clearly seen at night. In warm, humid climates like New Orleans, the warmth of the air is retained late into the night, finally cooling by morning. Water vapor and CO2 do that. In dry desert or arctic areas, the warmth of the day very rapidly is re-radiated into outer space and nighttime temperatures plunge rapidly. The absence of water vapor and CO2 do that. CO2 has the same effect as water vapor/humidity except in far less amount than water vapor/humidity. CH4 is even less than CO2.
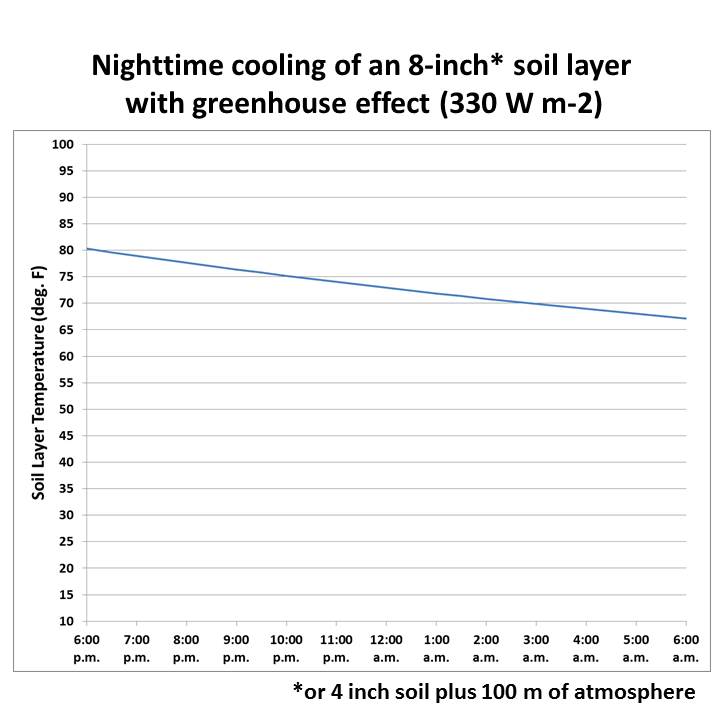
Figure 2
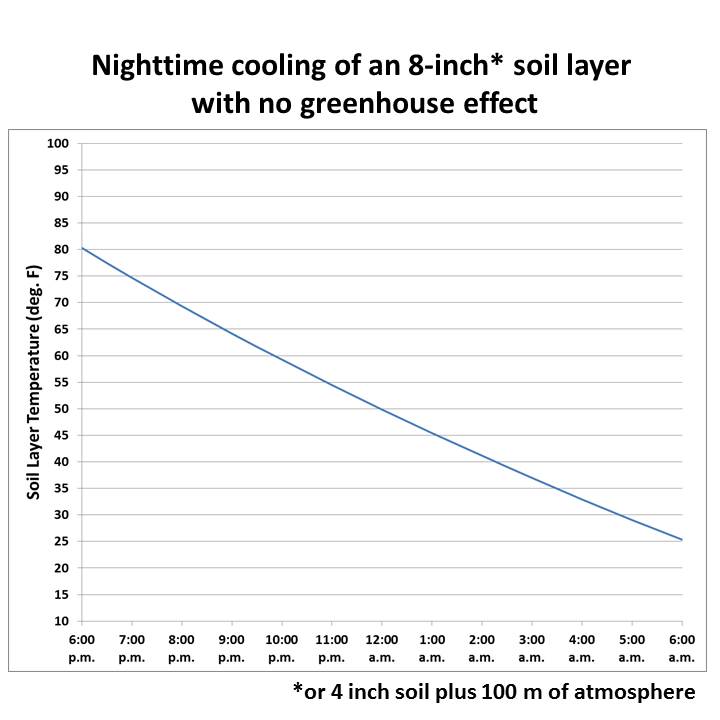
Figure 3
Nitrogen (78%), oxygen (21%) are the primary gases in the atmosphere, but these gases are mostly transparent to direct incoming solar radiation. Transparent means there are very limited or no energy bands available in these gaseous molecules to absorb energy at the wavelengths of the incoming solar radiation. A significantly large amount of solar energy is reflected by earth’s clouds, land and water surfaces back into outer space; this effect is known as albedo. The other atmospheric gases in total comprise about 1% of the atmosphere; this 1% includes water vapor, the inert gases (mostly argon which makes up 0.93%) and the trace gases. Carbon dioxide, methane and all other trace gases combined make up only about 0.04% of air.
Most radiant energy of the sun is transmitted through the gases in the atmosphere to the surface, i.e. to water and land surface, where it is absorbed. Most solar radiation is in the higher energy infrared, visible and ultraviolent wavelengths. Visible light, UV, X-Ray, and gamma radiation are all higher energy (higher frequency/shorter wavelength) than IR. Water vapor absorbs at certain visible energy bands…so we see darkening clouds as water vapor concentration increases. Solar radiation is absorbed by the molecules at the surface of water and land and then is spread through conduction and convection and dissipated in the energy cascade in the molecules of the water or solid matter. Some energy is re-emitted from the surface of land and water into the atmosphere but at lower energy, longer wavelengths, lower frequency than which it was originally absorbed.
Solar radiation received by the earth is known as Total Solar Irradiance (TSI). TSI has a cycle involving multiple variables which I will not go into here except to point out that climate cycles on earth correlate well with the TSI cycle and also with ocean temperature cycles known as PDO.
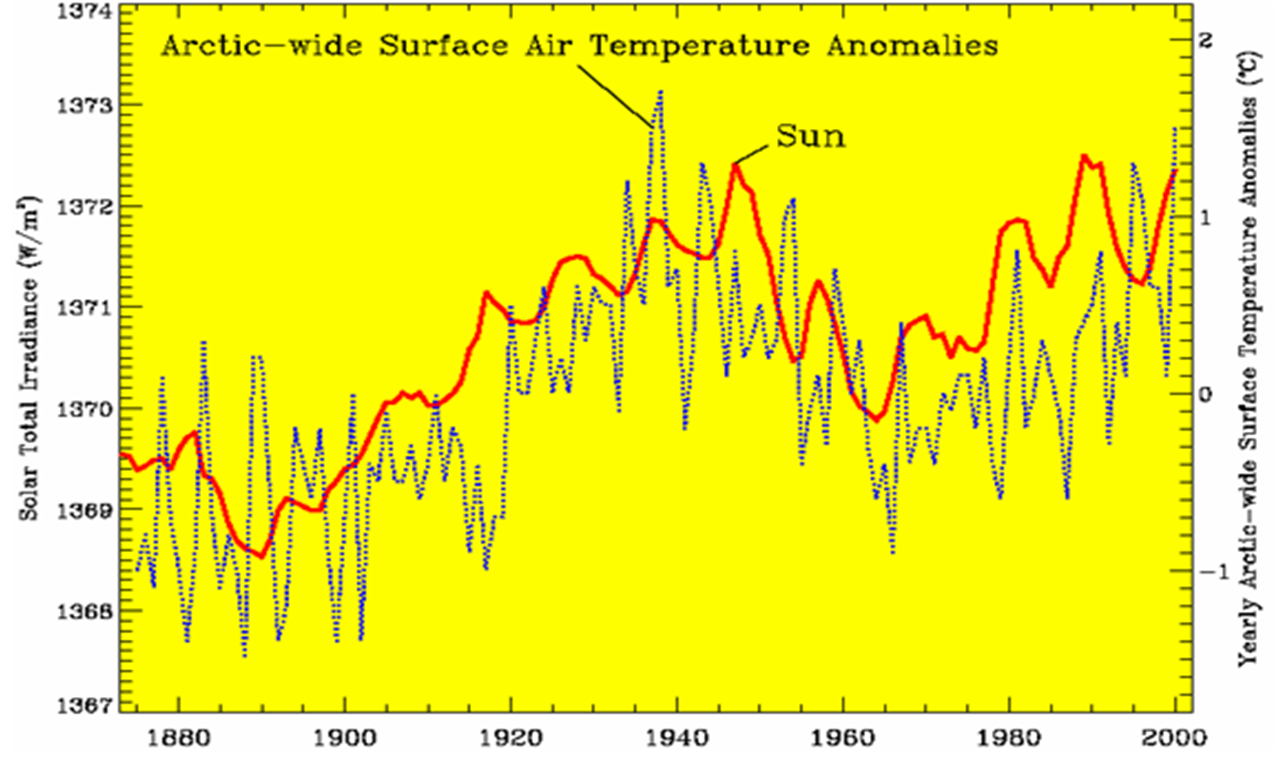
Figure 4
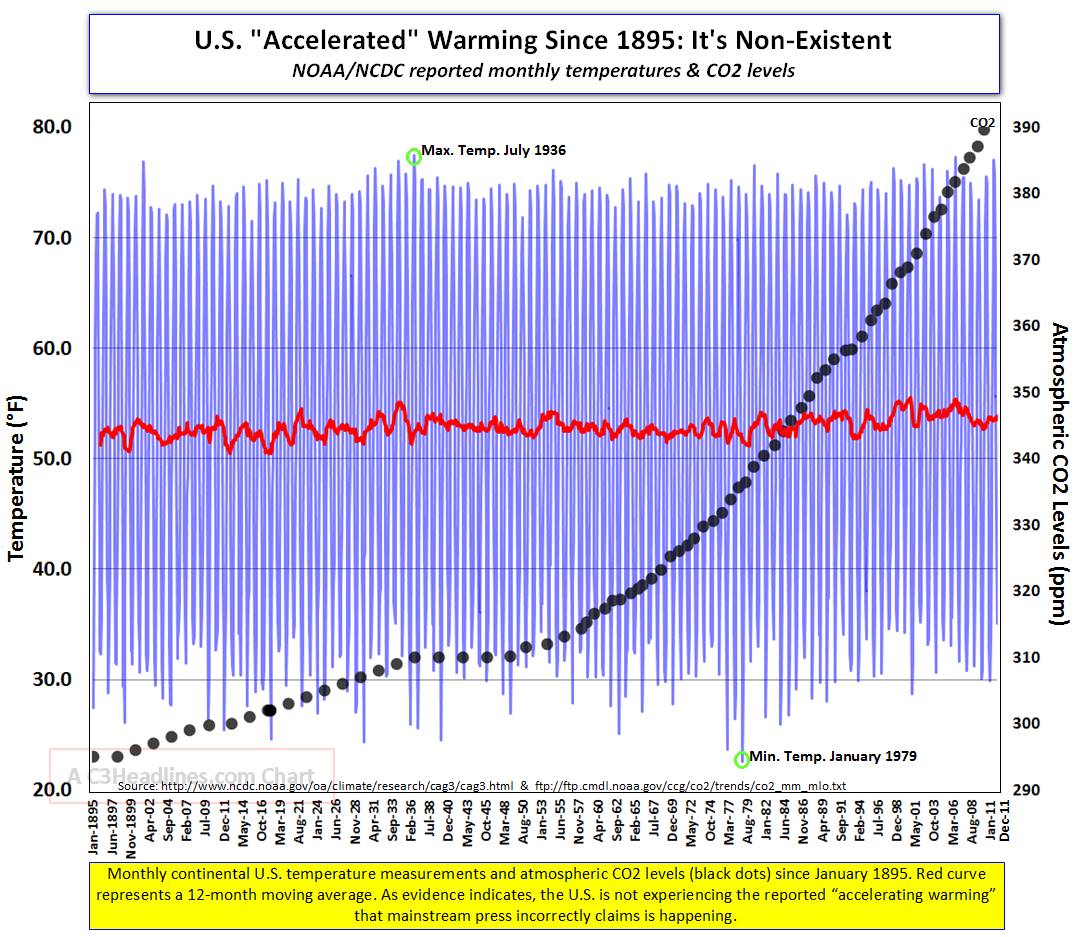
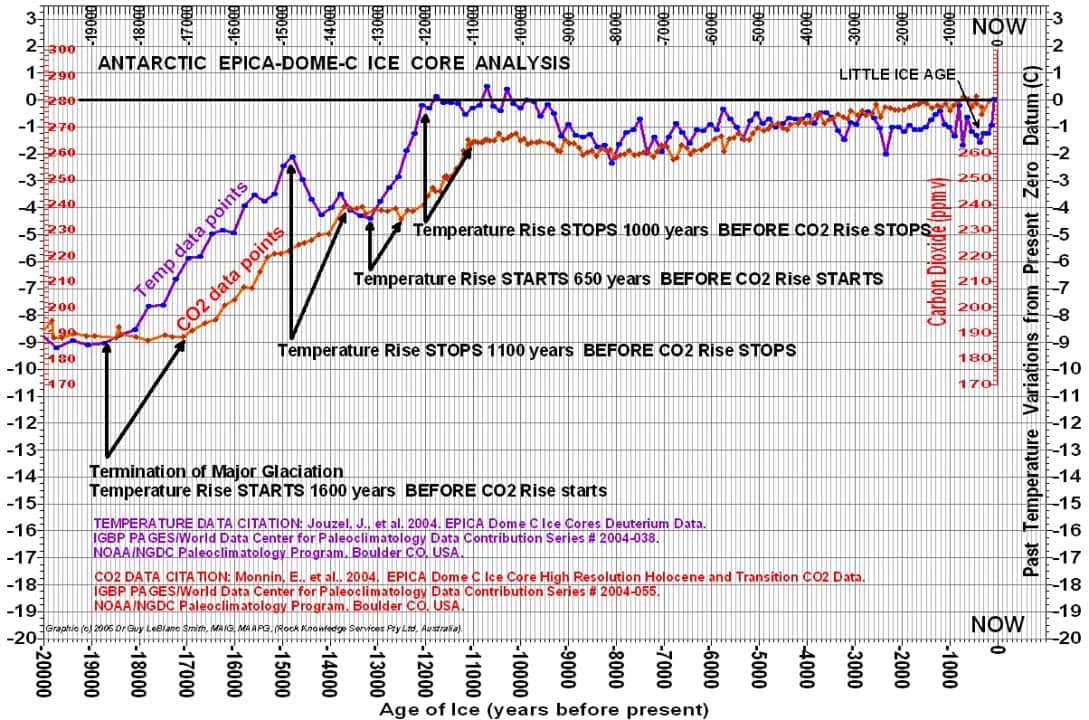
Temperature does not track CO2 very well (figure 11 below). Note temperature is flat, increasing, declining, and increasing while CO2 is increasing. Statistical correlation is weak.
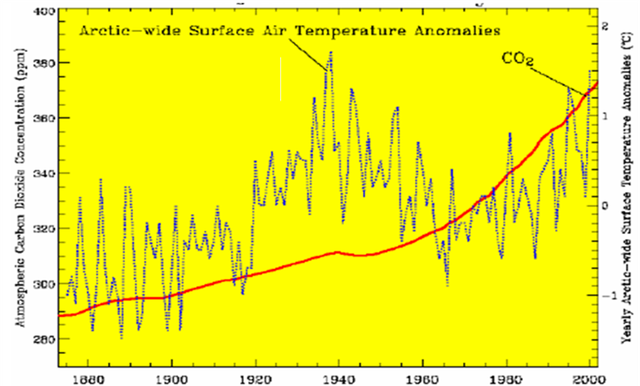
Figure 5
Over longer time periods, the correlation between CO2 and temperature is very poor. The two trends are widely diverging as seen in the graphic below. If the global warming hypothesis were valid, these two trends would be parallel or converging.
For the remainder of this discussion, I will consider only energy in the infrared (IR) wavelengths. Molecules in water and land absorb IR at several discrete higher energy/shorter IR wavelengths that match discrete energy bands of the electrons of each molecule. If the incoming energy does not match an energy band of the molecule, or if that energy band is not available to absorb because the band is already occupied with energy, then the energy passes through the molecule without being absorbed. If the IR energy is absorbed, then in a fraction of a second, part of that energy is dissipated as kinetic energy (various motions such as stretching, bending, and vibrations internal to the molecule or collisions with other molecules) leaving slightly lower energy/slightly longer wavelength/slightly lower frequency IR energy. That lower energy is then re-emitted. Since part of the energy has already been dissipated internally by the molecule, the emission has a longer wavelength, lower frequency of IR than the IR that was originally absorbed. Energy is not stored in a molecule except in the macro sense of all molecules taken together, and then only temporarily while the energy is progressively dissipated. Energy moves from one molecule to the next and at each step the energy is reduced. By this process, IR energy is absorbed by a water vapor, or CO2 molecule, or CH4 molecule for only fraction of a second and then re-emitted at a longer wavelength (lower frequency, lower energy, cooler). This cascade continues progressively downward in the energy until eventually the remaining energy is dissipated as kinetic energy by collision with other molecules, or various internal molecular motions.
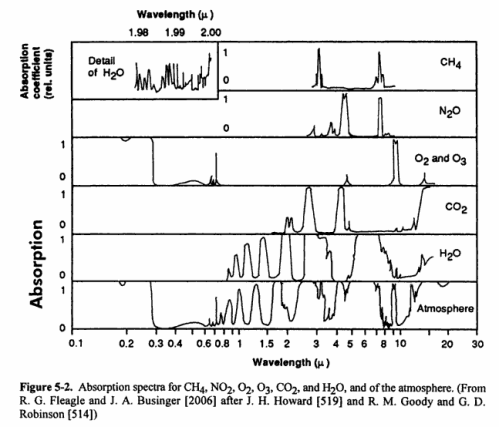
IR radiation is a sub-spectrum within the overall solar radiation spectrum. IR is 49.4% of solar energy reaching the outer atmosphere of earth. H2O, CO2, and CH4 absorb IR radiation ONLY at specific, discrete wavelengths (or quanta, or energy bands). Other wavelengths, which include most down-dwelling IR radiation directly from the sun, pass transparently through the molecules. Water vapor does absorb some incoming direct IR radiation. Some H2O, CO2, CH4 absorption wavelengths overlap (see graphic Figure 5-2), that is, these three molecules absorb IR at some of the same discrete wavelengths. This means that the gas which has the highest concentration in air absorbs and re-emits the most IR radiation. On average, water vapor including clouds is the highest concentration among the greenhouse gases, and thereby water vapor including clouds is the dominant greenhouse gas.
Most direct solar IR passes through air molecules to interact with molecules on the surface of water or land. Thus, infrared radiation is largely responsible for warming Earth’s surface, both land and water. In other words, the sun is not DIRECTLY warming the atmosphere, but INDIRECTLY warming the atmosphere. Most solar energy is absorbed in the molecules of the ocean and land which in turn warm the atmosphere. Most greenhouse warming is caused by reflected IR and IR emitted after dissipation from higher energy visible and UV energy. We don’t perceive visible light as heat; but when visible light interacts with matter, unless reflected, its energy is dissipated step by step in the downward energy cascade as described above until it becomes longer wavelength IR which then we perceive as heat. Re-radiation, conduction and convection from the surface of water and land warms the atmosphere.
IR energy only penetrates a few meters beneath the surface in liquid water in the oceans and lakes, while visible light transmits many meters beneath the surface of water, progressively loosing its colors from red to eventually higher energy blue and finally no visible light at increasing depth. The higher energy, shorter wavelength, higher frequency UV, x-ray and gamma energy mostly transmits through liquid water and finally interacts with molecules in solid matter in land or sea floor, or minimally with inorganic molecules dissolved in ocean water. Molecules on land and the seafloor surface are irradiated by these higher energy wavelengths, absorbing this energy, or conducting it further, and then these molecules progressively dissipate their energy as described above to progressively longer wavelengths and lower energy, eventually to IR wavelengths. The energy cascade continues on down the energy spectrum to satellite, wireless, radio, TV, MRI wavelengths and finally dissipated as kinetic motion of the molecule itself or collision.
The earth and the atmosphere also reflect and emit energy back into outer space, mostly in the visible and IR bands. IR emission from the atmosphere, water and land into outer space occurs day and night and is measured by satellite. Earth is an open system, continuously re-radiating its energy (which originally came from the sun) back into outer space, that is, radiating from higher energy earth to the much lower energy outer space beyond our atmosphere.
The fact that earth’s temperature is relatively stable over long periods of time, millions of years in multiple geological periods, implies that there is a balance between incoming energy and outgoing energy. I won’t go further into this energy balance topic here.
Over the very long time period of millions of years leading to the present, the earth has been in a slow cooling trend and has had a slowly declining atmospheric CO2 concentration as indicated in these graphics; this is probably contrary to what you have been told and taught.
On the other hand, and probably what you have been taught, in the geological short term, that is since the Little Ice Age and Industrial Revolution, earth warmed and total CO2 concentration increased. However most of that warming occurred before the 1950’s and that period was prior to the highest CO2 emissions by humans due to use of fossil fuels. In other words, the warming that followed the end of the Little Ice Age had very low CO2 concentration, thus that CO2 could not have caused the warming that occurred. The warming that occurred since the end of the Little Ice Age is within the statistical variation of the much longer-term cooling trend. In geological terms, since there is year-round ice at both poles, our present period is part of an interglacial period within an ice age.
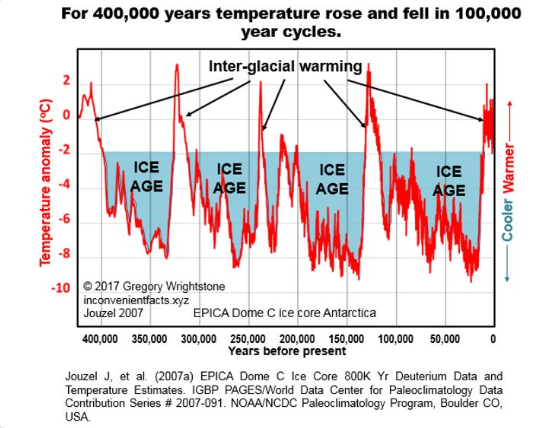
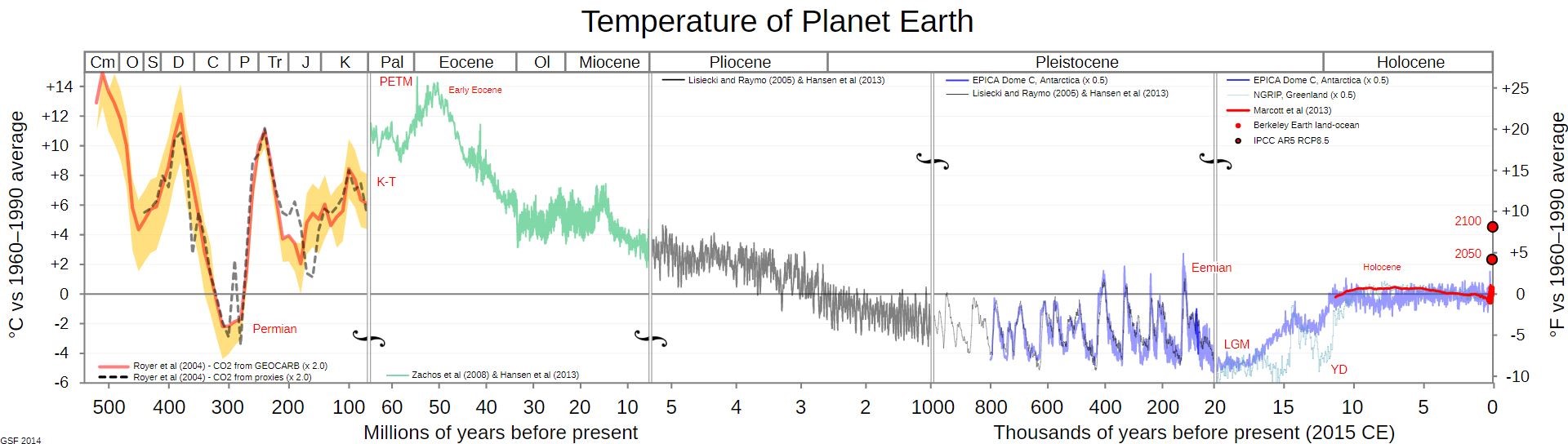
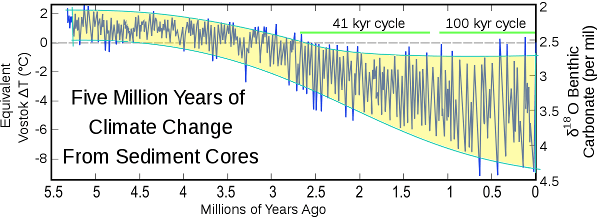
Figure 6
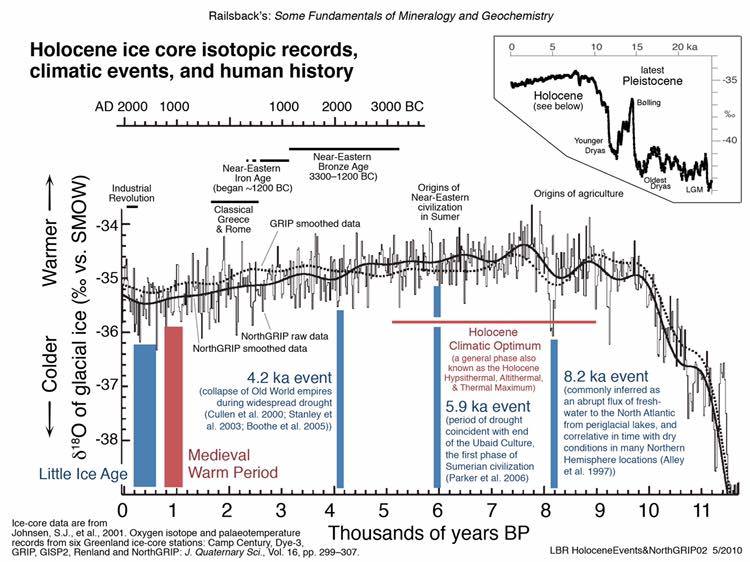
There has been global warming during our lifetime, mostly occurring before the 1950’s, but this warming is not unprecedented. And there has also been global cooling during our lifetime, also not unprecedented.
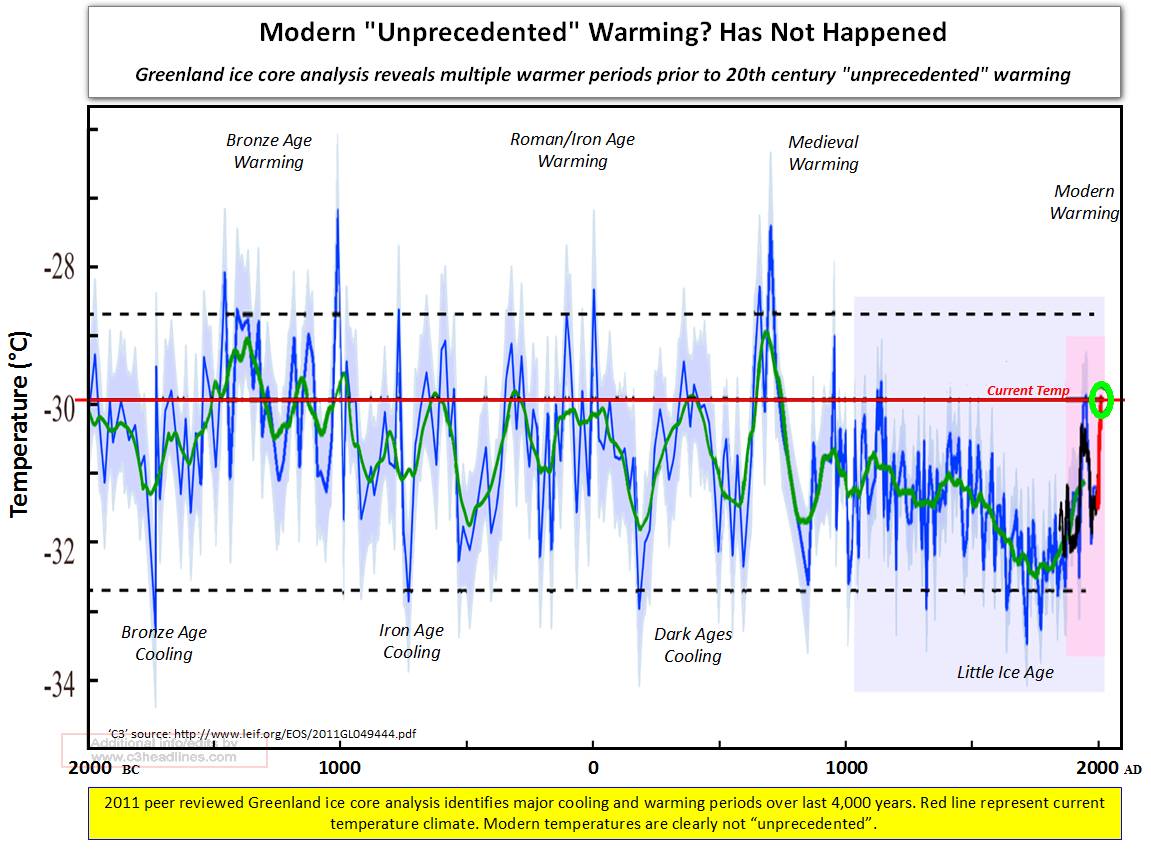
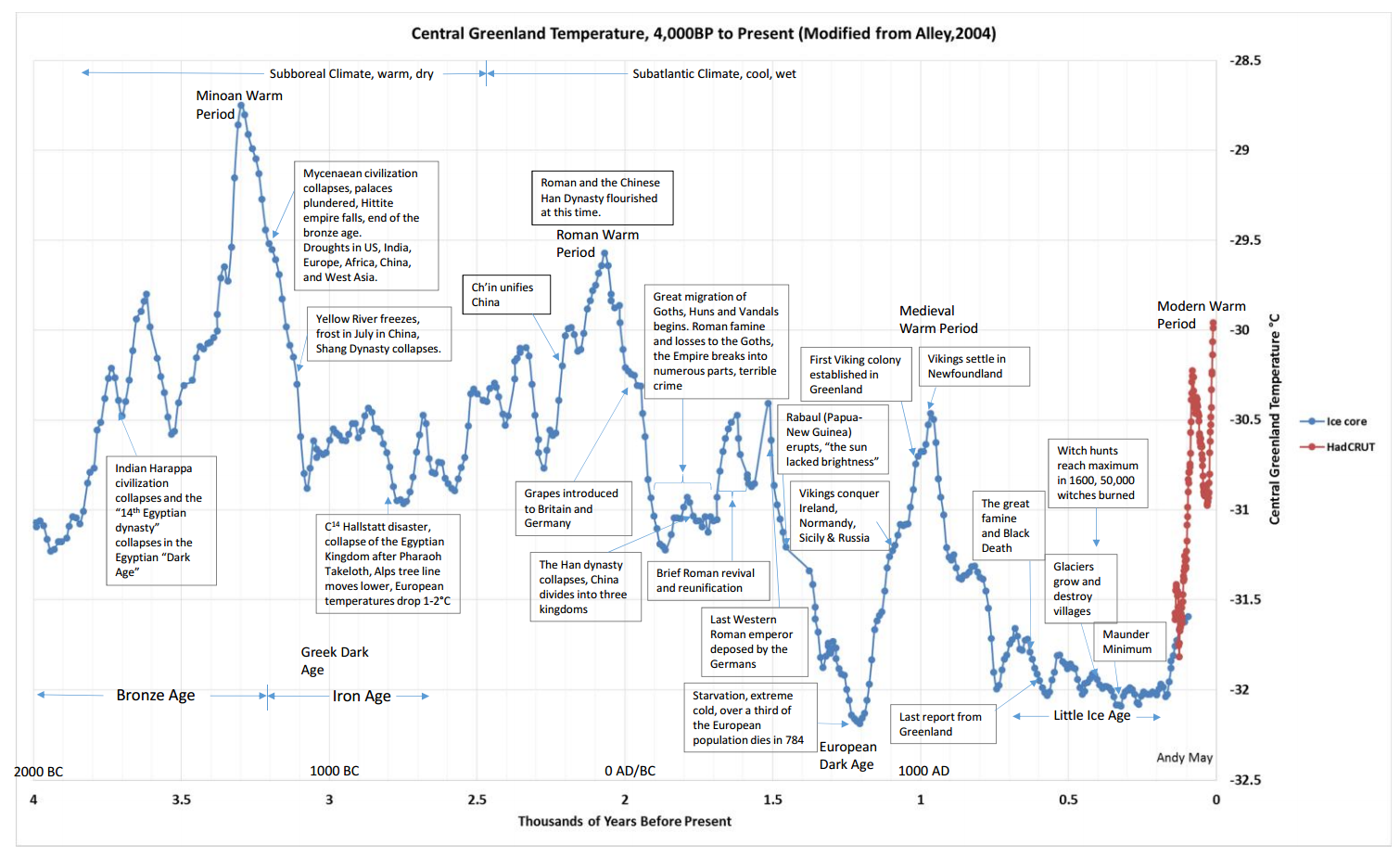
Figure 7
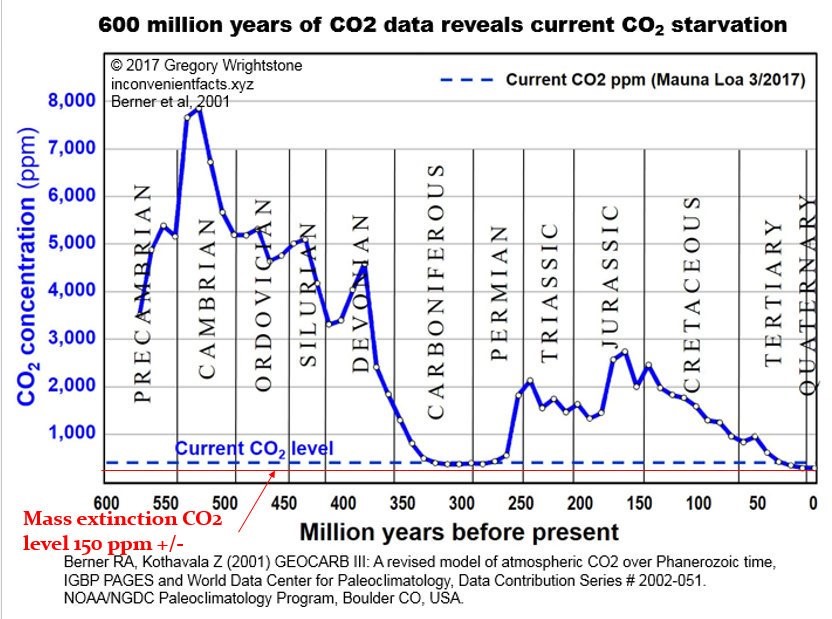
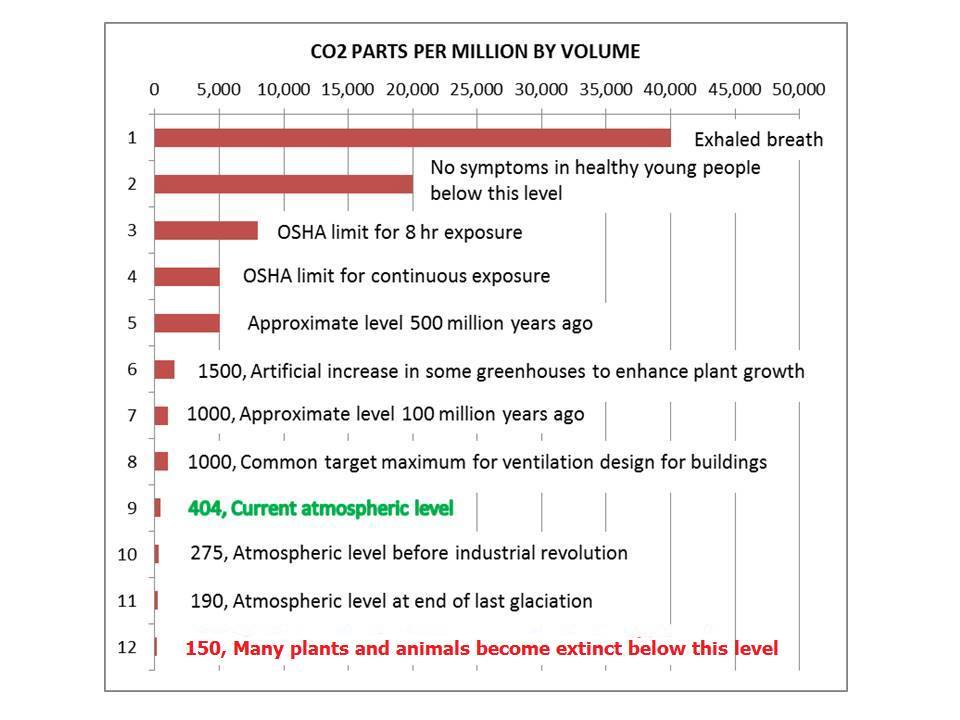
Figure 8
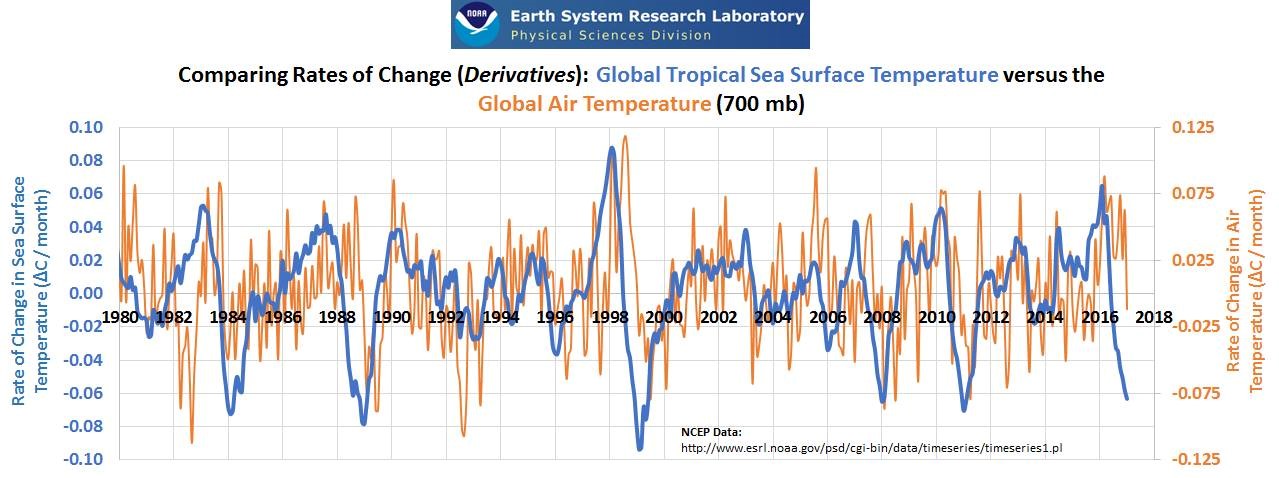
Returning to our “greenhouse gases,” the takeaway point is, again, the sun heats the earth, then the earth heats the atmosphere. The atmosphere is not heating the earth. According to the laws of thermodynamics, energy only flows from higher energy earth to the lower energy atmosphere. Earth (oceans and land) heat the air, not the reverse. Where can we measure this effect in the natural world? Detrended (i.e. seasons removed) sea surface temperatures versus air temperatures, we see in the graph below that increasing air temperature ALWAYS FOLLOWS increasing sea surface temperature. No matter how high the CO2 or methane concentrations are in the air, the ocean temperature will control the air temperature of the earth. In bulk, like insulation, water vapor, CO2, and methane temporarily delay but do not halt the continuous dissipation of energy/heat via the process described in the above paragraphs.
According to the proposed hypothesis for human-caused global warming (AGW), the continuing accumulation of CO2 in the atmosphere causes or forces increased warming in the atmosphere. The hypothesis requires disregarding the important facts that warming has currently stopped and also that there has been cooling for some periods in our lifetimes and historically, when according to AGW there should have been an increasing rate of warming due to the increasing concentration of CO2. Also, again falsifying the AGW hypothesis, when warming periods have occurred, there is no difference in the rate of warming during those periods despite the fact that CO2 has increased.
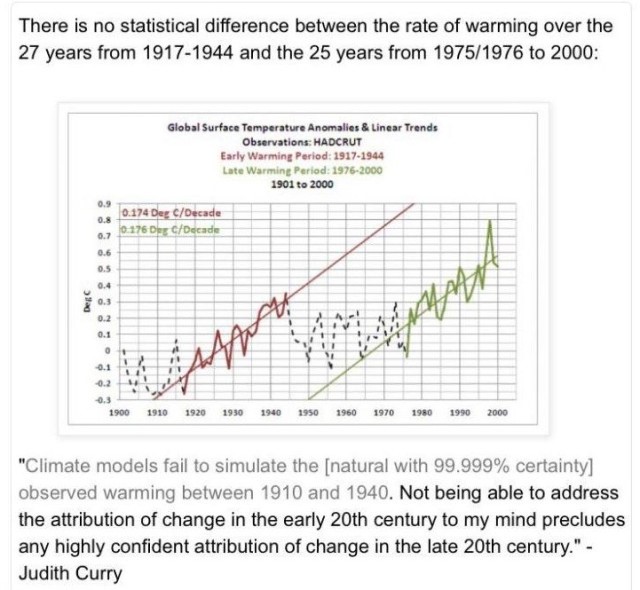
Figure 9
Based on a single molecule, one CH4 molecule absorbs more energy and re-emits more energy than one CO2 molecule, and one CO2 molecule absorbs more energy and re-emits more energy than one H2O molecule. But, the concentration (or abundance) of water vapor in air is about 1%, while the concentration of CO2 in air is only about 0.04% (or 400 ppm) and the concentration CH4 in air is only about 0.0018% (or 1.8 ppm). Thus more water vapor molecules in the air absorb more IR energy upgoing from the earth. Also, the IR radiation absorption wavelengths of water vapor overlap most of the IR radiation absorption wavelengths of CO2 and CH4. Since the concentration of water vapor is more than 10 times higher than that of CO2 and 100 times higher than CH4, the IR radiation emitted by the ocean, land and sun is more than 10 times more likely to be absorbed by water vapor molecules than by CO2 molecules and 100 times more likely to be absorbed by water vapor molecules than CH4 molecules. Then, when the water vapor, CO2 and CH4 molecules re-emit their energy upgoing and horizontally, or else collide, once again these emissions or collisions are far more likely to be interactions with a water vapor molecule; these phenomena change with increasing altitude.
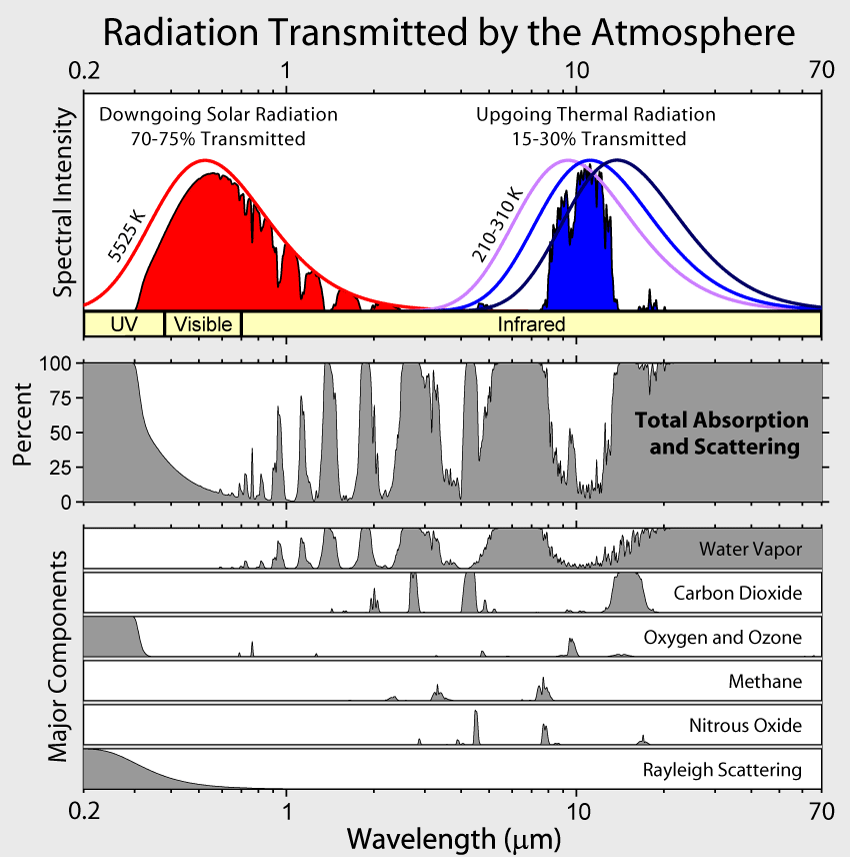
Figure 10
With regard to CO2 and CH4, the net result is that exponentially less warming results from each incremental increase in CO2 or CH4 concentration. The natural logarithm function is the inverse of the exponential function. The graphic below illustrates the natural log curve of temperature versus CO2 concentration that results from progressively increasing the CO2 concentration. In climatology terms, climate and temperature sensitivity are progressively diminished as CO2 concentration increases. Each doubling of CO2 concentration results in incrementally less warming, following a logarithmic progression. The available quantum energy bands of CO2 and CH4 are already occupied with energy received from nearby molecules. Adding more absorbing molecules decreases the mean-free-path of the radiation between molecules. Progressively higher levels of CO2 (and methane) produce progressively less warming. The absorbance of radiation is logarithmic.
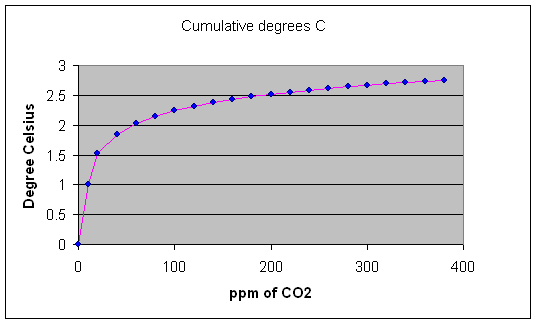
Figure 11
Furthermore, of the approximate 400 ppm (or 0.04%) total CO2 concentration in earth’s atmosphere, humans from all sources emit only about 3% of that total. About 97% of atmospheric CO2 is natural. About 98.5% of total CO2 is absorbed by nature.
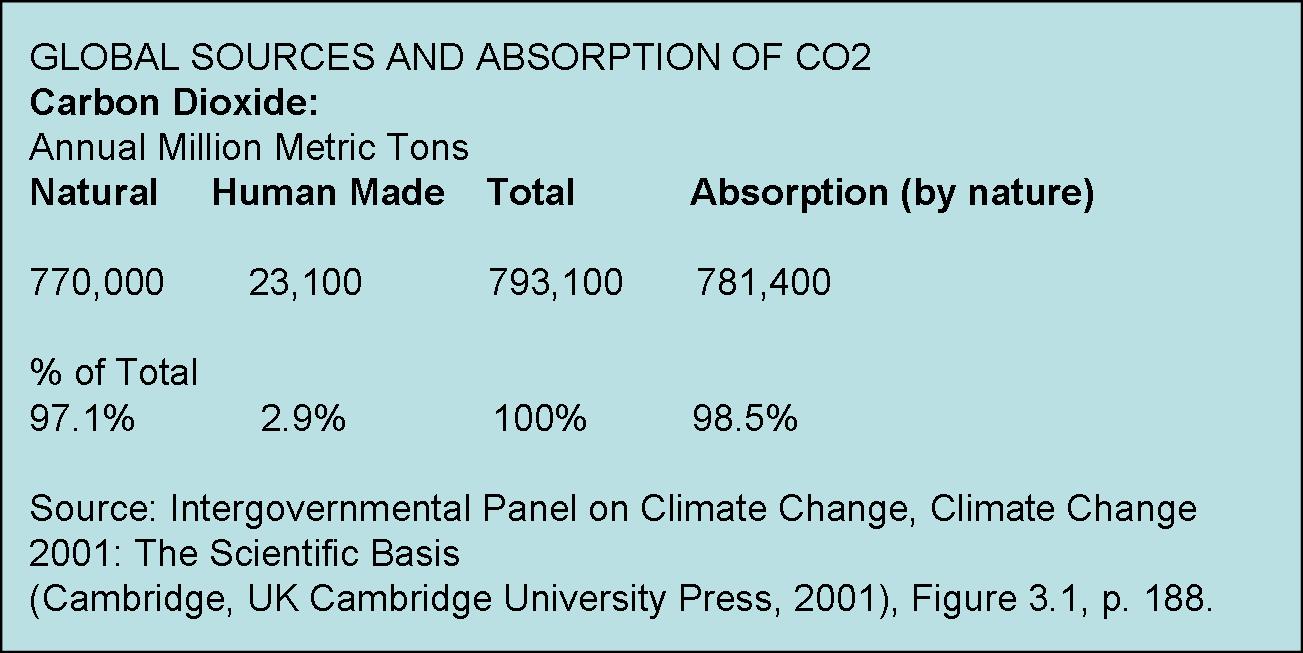
CO2 concentration has been increasing since the end of the Little Ice Age, around the time of the Industrial Revolution, primarily because earth has been slowly warming since that time. The human contribution is minimal. But, since CO2 is not causing significant warming (or even reproducibly measurable warming), and since the earth is in fact becoming greener according to NASA satellite studies, then there is no good reason to reduce CO2, or to reduce use of fossil fuels. Also, since most CO2 is natural, there is no practical way for humans to significantly reduce atmospheric CO2 concentration. Carbon sequestration, carbon offsets, carbon taxes, carbon trading are environmentally useless, harmful, economically wasteful, fraud. On the contrary, there are benefits to higher CO2, such as increasing crop yields. And there are also benefits to warming.
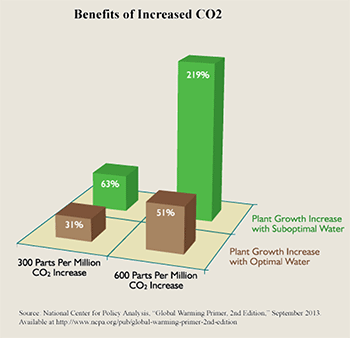

Figure 12
Despite clear evidence that the atmosphere is not significantly warming the planet, and under the laws of physics the atmosphere could not significantly warm the planet, still some politicians and scientists try to alarm citizens to persuade them that increasing CO2 and CH4 concentration forces catastrophically dangerous, large increases in temperature trends by increasing absorption of IR by water vapor. The alarmists’ torturous and unvalidatable hypothesis and models treat water vapor as part of a feedback system rather than as the dominant greenhouse gas. But, if this hypothesis were true, and it is not, since CO2 has been increasing steadily since the end of the Little Ice Age (CO2 increased about 150% from a very low CO2 level), then we should be seeing rapidly rising global temperature due to this feedback forcing effect, and we should be seeing a difference in rate of warming over time (or significant difference in second derivative.) However, the temperature trend has been only slightly increasing, and then only sometimes, and the warming trends are not exceptional or unprecedented in nature, and the overall temperature trend is diverging from the steadily rising CO2 trend.
If CO2 were the cause of warming, then the CO2 and temperature trends would be parallel or converging during the same time periods, and the CO2 increasing trend would occur before the warming trend; however, these correlations are not found in real world evidence unless the time periods are cherry-picked or the data has been manipulated. Even worse for the climate alarmists, there are multiple time periods, decades, where the temperature trend decreased while CO2 trend increased…for example the 1960s to late 1970s when most scientists and news outlets were proclaiming global cooling. CO2 cannot force both warming and cooling.
Climate catastrophe and gloom are entirely based on computer models, models which have never been validated against scientific observations.

Cover of Time magazine, January 31, 1977.
As of December 2018, the global temperature measured by satellites (the most accurate and reproducible measurement available), is only 0.45 degrees F above the average temperature computed since 1978 when satellite measurement began, meanwhile CO2 has been steadily rising during this same period. That’s not much warming; but the CO2 concentration is growing. In other words, nature has falsified the global warming hypothesis and climate models. Temperature has not been increasing in unison in correlation with increasing CO2. https://www.nsstc.uah.edu/climate/2018/december2018/GTR_201812Dec_1.pdf
Importantly, as shown in the graphic below, trends of increasing temperature precede trends of increasing CO2. Many peer-reviewed scientific papers confirm this observation. If CO2 were the cause or were forcing temperature increases, then that CO2 increase must precede the trend of increasing temperature; unfortunately for the AGW hypothesis and climate alarmists that evidence is not observed. Obviously, an effect cannot precede its cause. In other words, earth’s temperature is controlling CO2 concentration, not the other way around. (Al Gore’s science fiction movies show CO2 increases preceding or coincident with temperature increases leading up to a runaway ‘hockeystick’ warming.)
Most CO2 is emitted by the oceans and lands, naturally, by degassing caused by the slow warming trend since the Little Ice Age. Warmer water holds less CO2 than cold water. The relative partition ratio of a gas such as CO2 between water and air is controlled by Henry’s Law. Compared to the atmosphere, the oceans contain enormous energy (as already shown). Oceans contain about 50 times more CO2 than the atmosphere and 20 times more CO2 than the biosphere on land.
It would take 10,000 years for humans to change the temperature of the ocean by a mere 1 degree even if humans devoted all energy resources full time to that effort. The effects of the oceans on the earth’s climate are enormous, but the warming effect of human CO2 is trivially tiny.
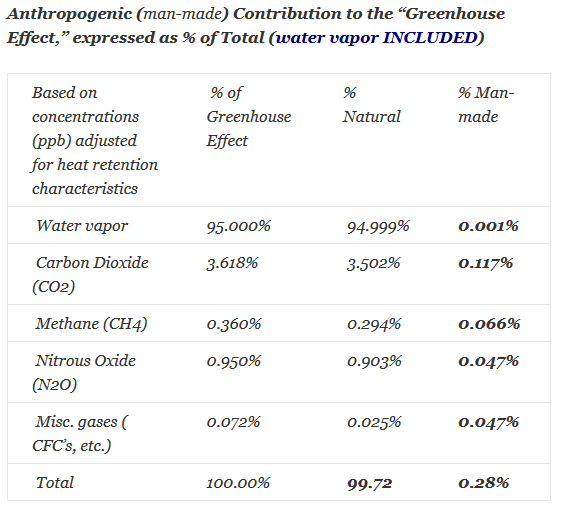
Figure 13
The claim that the relatively tiny amount of CO2 contributed to the atmosphere by humans is controlling earth’s temperature is falsified according to the rules of science. We mere humans are not able to change the temperature of earth by adding or subtracting CO2, no matter the amount of CO2. Even if the air were 40% CO2, an amount that is 1000 times higher than today’s total CO2 and 10,000 times higher than the human contributed CO2, the atmosphere can not significantly warm the oceans. Oceans are a gigantic heat sink or reserve, moderating the earth’s temperature and controlling earth’s climate.
This is NOT to say that humans do not affect temperature and climate in other ways, such as building cities, roads, planting or cutting forests and fields, etc. This IS to say that AGW, the hypothesis of human-caused global warming by the contribution of a mere 0.004% of the total CO2 emissions, is scientifically wrong. The human contribution to global warming is statistically insignificant, so tiny it is unmeasurable in the real world; the hypothesis of human-caused global warming/climate change has been falsified by real world observations and experiments.
What is really going on?
António Guterres, the current Secretary General of the UN, is the former president of Socialist International, which “was formed in London on 28 September 1864 by socialist, communist and anarchist political groups and trade unions. Tensions between moderates and revolutionaries led to its dissolution in 1876 in Philadelphia. The Second International was formed in Paris on 14 July 1889 as an association of the socialist parties.” (Wikipedia) With member organizations in more that 100 nations, following the communist tradition of Karl Marx, Leon Trotsky and Vladimir Lenin, Socialist International views the chief source of oppression and exploitation in the world to be capitalism — private property — whose inevitable by-products are poverty, environmental degradation, war, famine, and most other ills that plague humanity. UN leaders have also announced the UN’s anti-capitalist intentions. http://www.socialistinternational.org/viewArticle.cfm?ArticleID=1902
Bottom line: the global warming / climate change agenda is a giant, global fraud. The where, why, who, and when proponents knew it was a fraud is a subject for another day and a court of law. The global warming/climate change agenda is a global, political scheme to re-distribute wealth, change national economic systems away from capitalism, reduce national and individual sovereignty and reduce citizen control of private property, and hand over funding and control to unelected supra-national alliances and organizations like the UN and the EU.
References:
- https://www.omicsonline.org/open-access/New-Insights-on-the-Physical-Nature-of-the-Atmospheric-Greenhouse-Effect-Deduced-from-an-Empirical-Planetary-Temperature-Model.pdf
- Figure 2 and 3. http://www.drroyspencer.com/2015/04/why-summer-nighttime-temperatures-dont-fall-below-freezing/?fbclid=IwAR1Rv6gpWDE57LYYfeFSPQLa1tDIL7LfMQIoQhMrH0YDaiwSdGWDayoRMSA
- IBID
- Figure 4 and 5. Dr. Willie Soon. https://wattsupwiththat.com/2011/09/16/arctic-ice-refreezing-after-falling-short-of-2007/ and https://agupubs.onlinelibrary.wiley.com/doi/full/10.1029/2005GL023429
- IBID
- Figure 6 Lisiecki and Raymo (2005) http://what-when-how.com/global-warming/five-million-years-of-climate-change-from-sediment-cores-global-warming/
- Figure 7. Kobashi, T., K. Kawamura, J.P. Severinghaus, J.-M. Barnola, T. Nakaegawa, B.M. Vinther, S.J. Johnsen, and J.E. Box. 2011. High variability of Greenland surface temperature over the past 4000 years estimated from trapped air in an ice core. Geophys. Res. Lett., 38, L21501, doi:10.1029/2011GL049444. ftp://ftp.ncdc.noaa.gov/…/iso…/gisp2-temperature2011.txt
- Figure 8. https://pbs.twimg.com/media/DeuAH_4VMAAlBDd.jpg
- Figure 9. https://wattsupwiththat.com/2014/09/03/laughable-new-paper-claims-99-999-certainty-global-warming-over-past-25-years-is-man-made/
- Figure 10. https://wattsupwiththat.com/2011/02/28/visualizing-the-greenhouse-effect-atmospheric-windows/
- And https://thebestschools.org/special/karoly-happer-dialogue-global-warming/happer-major-statement/
- Figure 11. http://employee.heartland.edu/rmuench/carbondioxide.htm
- Figure 12. Sherwood Idso, PhD. http://www.co2science.org and https://thebestschools.org/special/karoly-happer-dialogue-global-warming/happer-major-statement/ Dr. Sherwood Idso with Eldarica pine trees grown in various amounts of CO2 in experiments done about 10 years ago when the ambient concentration of CO2 was 385 ppm. S.B. Idso and B.A. Kimball, “Effects of Atmospheric CO2 Enrichment on Regrowth of Sour Orange Trees (Citrus aurantium; Rutaceae) after Coppicing,” American Journal of Botany, 1994, 81: 843–846.
- Figure 13. https://www.geocraft.com/WVFossils/greenhouse_data.html Table 4a.
- 14. Sorokhtin et al. (2007) Sorokhtin, O.G., Chilingar, G. V., Khilyuk, L.F. (2007). Global Warming and GlobalCooling. Evolution of Climate on Earth,Developments in Earth & Environmental Sciences 5, Elsevier, ISBN 978-0-444-52815-5.
- 15. Climate Change Reconsidered. http://climatechangereconsidered.org/#tabs-1-2
- 16. https://budbromley.blog/2019/01/20/climate-impact-of-increasing-carbon-dioxide/
This article is re-published here:

Pingback: There is not any Local weather Emergency ! – All My Daily News
Oh-Two
I can’t live without you
Too much, you burn me up
Too little, I gasp for more
Twenty-one percent
Of all the air I breathe is you
I need trees too, ‘cause
Trees make oh-Two
And trees need you
Combined with “C” in C-oh-Two
So, I need C-oh-Two too
But too much C-oh-Two
Is bad, they say
Zero point-oh-four percent
Of the air I breathe
Is C-oh-Two
Would less C-oh-Two be
Enough for the trees
To make enough Oh-Two for me?
LikeLike
Nice work Ron. I shared widely.
LikeLike
Pingback: What if There is no Climate Emergency ? | US Issues
Pingback: What if there is no Climate Emergency ? – Weather Brat Weather around the world plus
Pingback: NZ’s Zero Carbon Bill: the Lies of Jacinda Ardern – Sto Vounó
Pingback: What If There is No Climate Emergency? | US Issues
I think Pat Frank’s study in uncertainty propagation in GCMs should be mentioned here.It reinforces your skepticism towards climate model accuracy:
https://www.frontiersin.org/articles/10.3389/feart.2019.00223/full
LikeLiked by 1 person
Thanks for your comment, reference and link. Excellent. Pat Frank’s article was discussed at WUWT, but I am glad to have the full paper.
More than 10 years ago, Roy Spencer explained in detail with examples why the climate models were wrong. All models were overstating climate sensitivity to greenhouse gases. The models were all (at the time) too sensitive to radiative forcing because they had misunderstood feedback processes, that is, feedbacks are an effect of radiative forcing, not a cause. His blockbuster revelation was that ALL feedback from radiative forcing is negative. Satellite and Climate Model Evidence Against Substantial Manmade Climate Change, by Roy W. Spencer, Ph.D. December 27, 2008 (last modified December 29, 2008)
https://www.drroyspencer.com/research-articles/satellite-and-climate-model-evidence/
LikeLike
Pingback: KOMMONSENTSJANE – CO2 IS NOT CAUSING GLOBAL WARMING. | kommonsentsjane
Reblogged this on Climate- Science.press.
LikeLiked by 1 person
Thanks Uwe. Danke. .
LikeLiked by 1 person
What climate data would change your mind? How might you know if you’re wrong?
I had to lay out exactly these answers when I was challenged back in the 90s and, like you, thought there was enough uncertainty to warrant doubt about AGW. My preconditions were not just met but continue to be exceeded. What about yours?
LikeLike
Thank you for your comment. I wish all climate scientists asked themselves such questions. That is how science is supposed to be done. That is how I looked at the question.
First, I would have to see statistically significant evidence that human-produced CO2 was increasing the growth trend in atmospheric CO2 concentration. Prof Salby and others have shown that the human contribution is insignificant. Since we already have solid evidence from Salby, Idso, Segalstad and others that the human component is insignificant, then before anything else the work of these scientists would have to be refuted.
Second, I would have to see evidence that CO2 was not following Henry’s Law.
Third, I would have to see evidence that the energy absorbed and transmitted by CO2 was not following the laws of thermodynamics with respect to a cooler object (a CO2 molecule) increasing the temperature of a warmer object (a molecule on earth’s surface.)
Fourth, I would have to see evidence of the atmosphere warming the oceans, including a testable explanation of the physical chemistry at the water’s surface. This evidence would have to somehow refute the abundant evidence that ocean temperature trends precede air temperature trends.
Fifth, I would have to see a repeated positive correlation over a millennium-long time period wherein a statistically significant trend of increasing temperature preceded a statistically significant trend increasing CO2. concentration.
Sixth and last but not least, I would have to see observable, testable evidence that CO2 causes statistically significant global warming over a multi-year period. So far, the only evidence I have seen is that CO2 causes a delay in time (or reduction in the rate) of cooling…i.e. CO2 functions as a insulator.
Thanks again for your comment and thanks for reading.
LikeLike
Yes, the scientific method and falsifiability is very important and on this we agree. But so, too, is probability and likelihood of modelling being correct when successfully applied. I see nothing about this in your list of what might convince you otherwise. The understanding of atmospheric CO2 we have today used to model AGW in fact produces tangible results consistently and reliably today in applications that work, in technologies that work. Right now. All the time. For everyone. Everywhere. That fact should reduce the doubt cast at our understanding of atmospheric CO2 and other greenhouse gas emissions, reduce the doubt that you are promoting, until you can produce a better one, an alternative explanation that accounts for why these applications and technologies work in spite of what you say is our incorrect understanding of atmospheric CO2. And that means you’re just casting doubt.
Casting doubt at what works for everyone everywhere all the time is not very scientific but obviously ideological.
That lack of scientific accountability you demonstrate by not producing an alternative model of understanding that works better is or should be a great big red flag to anyojne scientifically literate. Without that alternate understanding, it is only reasonable to doubt the doubter because your motivation is not to correct and/or replace the scientific understanding but to push the doubt aspect only.
This is why I asked what could convince you that you might be wrong, that AGW is in fact a increasing climate change driver? None of your list addresses in concrete terms how this could be done while keeping true to our current understanding of atmospheric CO2. This is the core problem here: you are insisting that our understanding of atmospheric CO2 is wrong so the AGW modeling is wrong. It is from reality that we have adduced our understanding (and then reverse applied to great success and reliability) and so you’re really asking reality to change to convince you otherwise!
The scientific understanding of atmospheric CO2 today is demonstrated with applications and technologies that work reliably and consistently well. And this matters tremendously.
You are arguing with all kinds of charts and graphs and scientific references that NASA’s understanding of how infrared energy through the atmosphere and the scientific basis of many instruments they utilize is factually wrong.
Why doesn’t – or shouldn’t – that give you pause?
You and, say, all the atmospheric scientists at NASA can’t both be right when you reach incompatible conclusions… yet the same instrumentation NASA uses based on NASA’s understanding of atmospheric CO2 is also relied upon for military use… and to great effect and accuracy in targeting. Are you going to double down here and insist they’re wrong, too, but continue to offer no alternative understanding… unless someone else (who also brought us our understanding of acid rain, who brought us our understanding of ozone depletion) produces “strong evidence that CO2 is not following Henry’s Law?”
See, Bud, that’s not reasonable doubt you’re blogging about, not scientifically responsible doubt. That’s just manufactured doubt blown out of proportion for ideological reasons dressed up to appear to be scientific. This is the proto-typical method used by anyone who wishes to undermine scientific results that inconveniently work for everyone everywhere all the time. For you to be correct, none of these applications or technologies should work. Yet they do when according to you and your charts and graphs and references so opaquely argue our modeled understanding is wrong.
So it is in fact reality that is demonstrating to you that your level of doubt is unwarranted because there’s something wrong somewhere in all those contrary charts and graphs and references that leads you to this irrational doubt while turning a blind eye to all the modeled understanding we use for applications and technologies that work reliably and consistently well in every other aspect of atmospheric science.
LikeLike
I don’t doubt the laws of physics, chemistry and math.
A computer model is only a hypothesis. I have built many, but they were business and economics models not climate. But the principles are the same. A computer model is a hypothesis, nothing more. Models require validation by observations in the real world. If the model/hypothesis does not validate, then you start over. But the climate industry does not start over. They are not even asking the right questions in their models. They don’t understand and model the natural environmental variables, clouds and water vapor for example, but they plug in assumptions about human contributions. That is guaranteed to be a failed model.
As a matter of fact, ALL of the current computer models of climate are notoriously bad (except the Russian model.) They fail to accurately re-produce known climate conditions even when all of the variables in the model are substituted with known, measured values…they fail back validation against known conditions. In real science, that means the hypothesis is falsified and you go back to the drawing board. But AGW proponents are not doing that. John Christy at University of Alabama has testified extensively about this in Congress and elsewhere.
The models don’t work because they are built on mistaken assumptions. For example, models assume down-dwelling radiation from CO2 is warming the surface. That assumption defies LAWS of physics (not hypotheses or theories, but LAWS.) Another example, the models assume that CO2 in the atmosphere warms the oceans, also defying laws of physics and physical chemistry. Another example, the variables used for radiation and emission (Watts per square meter) assume flat surfaces (so that the algorithms fit other theories), but the surface of the earth and the upper boundary of earth’s atmosphere are not flat and that is important because of Beer’s Law. Another example, Henry’s Law determines the ratio of CO2 in the ocean versus the air above it and the ocean is an infinite sink for CO2, which means there can NEVER be an acidic ocean (pH lower than 7) due to CO2, and we can measure that. But how many times have you heard or read AGW alarmists preach about ocean acidification?
AGW proponents make up and falsify data that is easily disproven. For example: IPCC claims that the rise in CO2 to 353 ppmv in 1990 and 379 ppm in 2005 is due to anthropogenic CO2 (IPCC 1990, 2007).
• Stable 13C/12C isotope ratios, expressed as δ13C give us the only way to unequivocally determine the fraction of anthropogenic CO2 in the atmosphere. [This is a factual measurement made by carbon isotope ratio mass spectrometry … i.e. not a computer model or assumption.]
• The natural atmospheric CO2 reservoir has δ13C -7 when in isotopic equilibrium with marine HCO3- and CaCO3.
• CO2 from burning of fossil-fuel and biogenic materials has δ13C = -26.
• δ13C reported for atmospheric CO2 was -7.489 in Dec. 1978, decreasing to -7.807 in Dec. 1988 (Keeling et al. 1989).
• If the decreasing δ13C was only caused by mixing natural CO2 with CO2 from burning of fossil fuels or plants (79%/21% CO2 mix; lifetime 50-200 years; IPCC 1989), then the current atmospheric CO2 δ13C should be -11, i.e. much lower than the IPCC value.
• The mass of all fossil-fuel emissions in the current atmosphere was 30 GT C or less, i.e. maximum 4%, corresponding to an atmospheric CO2 concentration of 14 ppmv.
• Above bullets quoted from: EGC-07 Frontiers of stable isotope analysis for environmental science and biogeochemistry. Carbon isotope mass balance modelling of atmospheric vs. oceanic CO2 by Tom V. Segalstad, Natural History Museum, University of Oslo (Norway).
There is no ideology involved in my analysis; the ideology is with the AGW alarmists. AGW proponents have an agenda, not me. It is their obligation to prove their case and they have not done so in 30 plus years despite spending billions of dollars trying. I have no agenda. I have no political party. I have no employer. I own no stocks, bonds or any other ties to industry, media, etc. I understand the chemistry and physics, apply those laws and do the math with an open mind.
If human contributed CO2 is statistically insignificant to the growth rate in CO2 concentration, which is science proven and replicated, then human-produced CO2 cannot be a significant contributor to warming or any other CO2-dependent global climate variable. ALL other climate variables drop out/cancel each other.
This is not ideology, it is fact: you and millions of people worldwide have been sold lies in order to generate money and political power. The climate industry relies on unvalidatable computer models and they commit fraud by adjusting data, omitting data and facts, and using assumptions which are known to be wrong or misleading. Yet they keep pushing this fraud because that is where their funding comes from. Even the concept of “greenhouse gas” is a lie. The primary function of a greenhouse is to block circulation of air, that is, to block most convection. But so-called “greenhouse gases” do not have that function at all.
With all due respect, it is time for you to wake up to reality.
LikeLike
If NASA – and almost all of the major scientific organizations in the world – is wrong in their understanding of atmospheric CO2 as you try to portray with a gish gallup of what appears to be scientific understanding, and yet all their instrumentation used by all kinds of other geographical information services, industries, governmental departments, and national defense equipment works reliably and consistently well based on this supposedly incorrect understanding, how can you not grasp just how likely is chance you are wrong?
How can you – admittedly not a climate scientist – know better and presume to think that your grasp of math, physics, and chemistry exceeds almost every other working climate scientist and engineer who makes these working applications and technologies?
Or do you really believe you’re about the smartest guy around in comparison and one of the very few who are able to see differently and clearly where all these others fail to do so?.
Do you really think so many climate scientists are really that ill-informed… the same ill-informed scientists whose misplaced scientific expertise was still good enough to determine that acid rain and ozone depletion needed addressing and who helped reverse damaging practices that caused these effects… but – all of a sudden – when it comes to climate change suddenly their expertise is ill-informed, incorrect, and well behind your own?
Seriously, Bud?
LikeLike
Your appeal to authority is lost on me. It is non-scientific and non-logical, and it devalues any argument you might make.
There are multiple studies like this one. Here is the abstract and reference.
“Assessing human impacts on climate and biodiversity requires an understanding of the relationship between the concentration of carbon dioxide (CO2) in the Earth’s atmosphere and global temperature (T). Here I explore this relationship empirically using comprehensive, recently-compiled databases of stable-isotope proxies from the Phanerozoic Eon (~540 to 0 years before the present) and through complementary modeling using the atmospheric absorption/transmittance code MODTRAN. Atmospheric CO2 concentration is correlated weakly but negatively with linearly-detrended T proxies over the last 425 million years. Of 68 correlation coefficients (half non-parametric) between CO2 and T proxies encompassing all known major Phanerozoic climate transitions, 77.9% are non-discernible (p > 0.05) and 60.0% of discernible correlations are negative. Marginal radiative forcing (ΔRFCO2), the change in forcing at the top of the troposphere associated with a unit increase in atmospheric CO2 concentration, was computed using MODTRAN. The correlation between ΔRFCO2 and linearly-detrended T across the Phanerozoic Eon is positive and discernible, but only 2.6% of variance in T is attributable to variance in ΔRFCO2. Of 68 correlation coefficients (half non-parametric) between ΔRFCO2 and T proxies encompassing all known major Phanerozoic climate transitions, 75.0% are non-discernible and 41.2% of discernible correlations are negative. Spectral analysis, auto- and cross-correlation show that proxies for T, atmospheric CO2 concentration and ΔRFCO2 oscillate across the Phanerozoic, and cycles of CO2 and ΔRFCO2 are antiphasic. A prominent 15 million-year CO2 cycle coincides closely with identified mass extinctions of the past, suggesting a pressing need for research on the relationship between CO2, biodiversity extinction, and related carbon policies. This study demonstrates that changes in atmospheric CO2 concentration did not cause temperature change in the ancient climate.”
Article: The Relationship between Atmospheric Carbon Dioxide Concentration and Global Temperature for
the Last 425 Million Years
W. Jackson Davis
Environmental Studies Institute, Boulder, CO , 80301, , USA; acksonDavis@EnvironmentalStudiesInstitute.org
Division of Physical and Biological Sciences, University of California, Santa Cruz, CA, 95064, , USA
; Published: 29 September 2017
(PDF) The Relationship between Atmospheric Carbon Dioxide Concentration and Global Temperature for the Last 425 Million Years. Available from: https://www.researchgate.net/publication/320123470_The_Relationship_between_Atmospheric_Carbon_Dioxide_Concentration_and_Global_Temperature_for_the_Last_425_Million_Years [accessed Apr 30 2019].
LikeLiked by 1 person
First, the irony seems lost on you that your criticism of my comments rests on the accusation that I appeal to authority while rebutting me with a few selected studies that – you guessed it – appeal to authority!
Secondly, you misunderstand my central point: that the validation of the current understanding of atmospheric CO2 rests not in authority (as you accuse me of relying on and the means by which you produce a gish gallop of charts, graphs, and references) but in successful practical applications and technologies of that understanding that inconveniently work for everyone everywhere all the time. This is what refutes you: these applications and technologies. That is why to refute the consensus means you have to come up with not a few selected studies that appear to support doubt of our current understanding but a better explanation. And on this point, you have utterly failed.
LikeLike
You have not posted a single article or link. You have not offered any scientific argument. My original posts, both of them, as well as my replies to you do not appeal to authority. Do you even know what that is? Are you a bot? You have offered absolutely nothing.
LikeLike
Invalidity of EPA’s CO2 Endangerment Finding
Posted on February 5, 2019 by budbromley
Excerpted summary of this peer-reviewed science: “The analysis results invalidate EPA’s CO2 Endangerment Finding, including the climate models that EPA has claimed can be relied upon for policy analysis purposes. Moreover, these research results clearly demonstrate that once the solar, volcanic and oceanic activity, that is, natural factor,impacts on temperature data are accounted for, there is no “record setting” warming to be concerned about.”
Title: On the Existence of a “Tropical Hot Spot” & The Validity of EPA’s CO2 Endangerment Finding. Abridged Research Report. Second Edition.
Here is a link to the updated second edition of this peer-reviewed paper, along with a link to the press release that went out with it. The previous version was also posted on my blog.
Click to access ef-data-research-report-press-release-0418172.pdf
Click to access ef-data-research-report-second-editionfinal041717-1.pdf
LikeLike
The Climatological Significance of a Doubling of Earth’s Atmospheric Carbon Dioxide Concentration. Idso SB.1
1U.S. Water Conservation Laboratory, 4331 East Broadway, Phoenix, Arizona 85040
Abstract: “The mean global increase in thermal radiation received at the surface of the earth as a consequence of a doubling of the atmospheric carbon dioxide content is calculated to be 2.28 watts per square meter. Multiplying this forcing function by the atmosphere’s surface air temperature response function, which has recently been determined by three independent experimental analyses to have a mean global value of 0.113 K per watt per square meter, yields a value of </= 0.26 K for the resultant change in the mean global surface air temperature. This result is about one order of magnitude less than those obtained from most theoretical numerical models, but it is virtually identical to the result of a fourth experimental approach to the problem described by Newell and Dopplick. There thus appears to be a major discrepancy between current theory and experiment relative to the effects of carbon dioxide on climate. Until this discrepancy is resolved, we should not be too quick to limit our options in the selection of future energy alternatives.”
PMID: 17779615 DOI: 10.1126/science.207.4438.1462
https://www.ncbi.nlm.nih.gov/pubmed/17779615
LikeLike
Bud, I thank you for your efforts and explanations. I’ve been following the claims of Global Warming for decades now and have always been skeptical of those claims because they enrich the claimants. And, the observations have not ever agreed with the models which are used as predictors and cannot be. I also read sites and am always surprised that they are so certain and to the untrained scientific eye (mine), they seem so certain of their position and the errors of the deniers.
What to make of all this discrepancy? It’s like being a juror at a trial in which someone has to be wrong but, how to tell which side is lying? We’re all arguing about the future which I’m sure Yogi Berra pointed out hasn’t yet happened. So, we can’t know if we’re experiencing climate variability or AGW. Before we proceed further with governmental policies based on belief and not knowledge, we need to find a method of arriving at knowledge and having that become widely known. Otherwise, Gov. Inslee, AOC, IPCC and Gore will (probably) cause us significant damage. It’s been said that what’s believed turns out to be reality.
LikeLiked by 1 person
gsmullennix: Thanks for reading and thanks for your comments.
I wish everyone thought like you. By that I mean, as Ted Nordenhaug my favorite philosophy professor would say, “Thinking is hard work, so hard it has rarely been tried.” People, including me, prefer to do the things we like to do and not be bothered with the rest. If there’s an issue, we seek someone or thing that has the answer so that we don’t have to do the hard work of thinking. It’s interesting probably to a normal bell curve distribution of humanity that humans explore the moon, and plan to explore Mars and beyond. But, I would guess that the few people who are interested enough to do the calculations which would allow a spaceship from earth to land safely with the moon are beyond three standard deviations from the norm. Only a few of them. And it is a safe prediction that is true in many areas. As a result, the mass of humanity wants its information and work pre-digested, if you will. We are all too willing to rely on someone else to think for us.
And life has been that way for a very long time. It is probably part of the human condition.
You said, “So, we can’t know if we’re experiencing climate variability or AGW.” I disagree. We do know, at least some of us know. We know that AGW is too small to be measured, buried in the noise. We know that CO2, the claimed culprit, does not correlate well with several other variables when it should correlate if CO2 were a significant contributor. We know that seemingly endless numbers of CO2-based climate models generated by AGW proponents have failed to predict past, present and future climates, which suggests that CO2 is not involved. [The Russian model appears to be predictive in its few runs.] We know that the people involved in the climate models are conflicted by the funds they receive.
Math tools such as quantum resonance interferometry easily [by orders of magnitude] have the potential to extricate from the noise the minuscule human CO2 contribution. But the modelers who are funded will never use those tools because their use alone validates that the human signal is less than noise, and the inevitable result would disprove their hypothesis.
So, you see, this annoying behavior by obviously biased so-called “climate scientists” is an example of the normal human condition. They have their comfort space. They are protecting their castle.
I don’t think you are not arguing the technical point, ““So, we can’t know if we’re experiencing climate variability or AGW.” Instead, I am using this as an example in response to your next three sentences, with which I strongly agree, “Before we proceed further with governmental policies based on belief and not knowledge, we need to find a method of arriving at knowledge and having that become widely known. Otherwise, Gov. Inslee, AOC, IPCC and Gore will (probably) cause us significant damage. It’s been said that what’s believed turns out to be reality.” In fact, those people are already causing us severe damage, to the tune of billions of dollars wasted on this climate fraud.
The best, the tried and true, method of arriving at knowledge (as opposed to mere information, opinion and belief) is to take responsibility and think for ourselves. That is hard work in a too lazy world. To assist my own efforts in that regard, I turned off all mainstream media 13 years ago. It’s amazing how much time there is to think, and how much less interference there is. Instead of receiving information that is pumped at me, I have the time to seek answers to my questions and evaluate them.
And don’t fall for the trap where you think you must allow every comment on your blog or respond to every comment and criticism. That easily can be an enormous waste of your time. It’s your blog. So, make your points.
In my round about, beat around the bush, way I am encouraging you and your new blog. I will try to find some of Ted Nordenhaug’s writings, which I think you will enjoy, and post them on your blog if you wish.
LikeLike